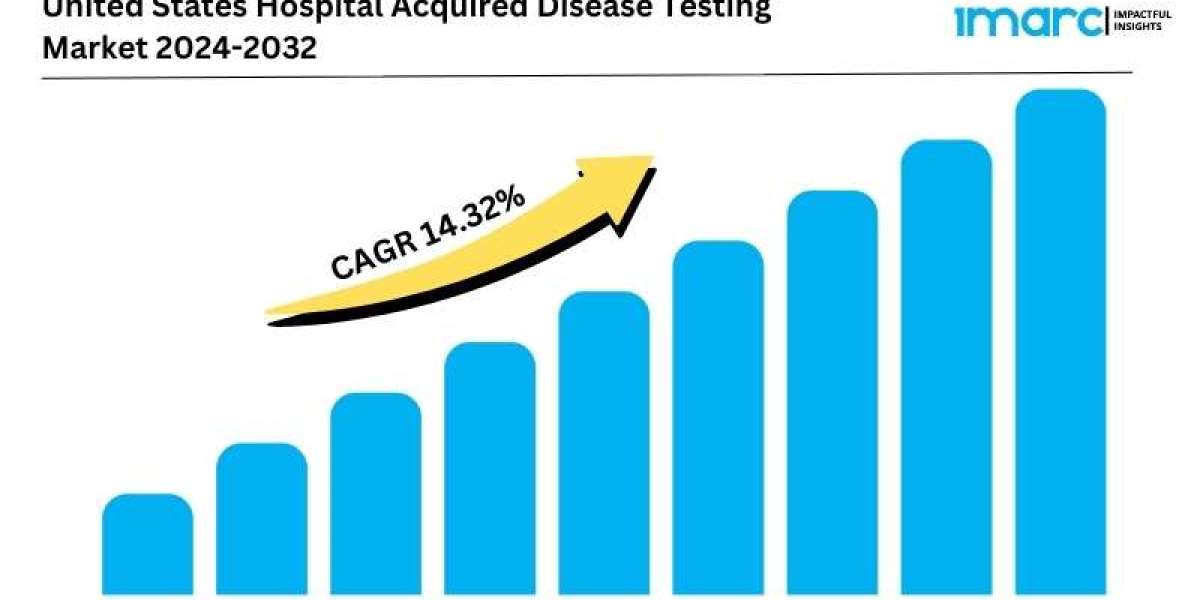
United States Hospital Acquired Disease Testing Market Overview
Base Year: 2023
Historical Years: 2018-2023
Forecast Years: 2024-2032
Market Growth Rate: 14.32% (2024-2032)
The United States hospital acquired disease (HAD) testing market is driven by the increasing prevalence of hospital-acquired infections (HAIs) and the stringent regulatory guidelines aimed at improving patient safety and care quality. According to the latest report by IMARC Group, the market is projected to grow at a CAGR of 14.32% from 2024 to 2032.
United States Hospital Acquired Disease Testing Industry Trends and Drivers:
HAIs, such as methicillin-resistant Staphylococcus aureus (MRSA), Clostridium difficile, and bloodstream infections, pose significant health risks and contribute to extended hospital stays, higher medical costs, and increased mortality rates. The Centers for Disease Control and Prevention (CDC) and other healthcare organizations have reported a steady rise in HAI incidences, prompting healthcare facilities to adopt comprehensive testing protocols to mitigate these infections. Regulatory bodies, including the Centers for Medicare & Medicaid Services (CMS), have established stringent guidelines and penalties for hospitals failing to reduce HAI rates. Compliance with these regulations necessitates regular and accurate testing for various pathogens, driving the demand for advanced diagnostic tests and technologies in the healthcare sector.
Technological advancements and the adoption of innovative diagnostic tools are also significant factors propelling the United States HAD testing market. The development of rapid, sensitive, and specific diagnostic tests, such as polymerase chain reaction (PCR) assays, next-generation sequencing (NGS), and automated microbial identification systems, has revolutionized the detection and management of HAIs. These advanced diagnostic tools enable quicker identification of pathogens, allowing for timely and targeted treatment interventions, thereby reducing the spread of infections within healthcare facilities. Additionally, the integration of artificial intelligence (AI) and machine learning (ML) in diagnostic testing is enhancing the accuracy and efficiency of HAI detection. AI-powered platforms can analyze vast amounts of data, identify infection patterns, and predict outbreaks, supporting proactive infection control measures. The growing emphasis on precision medicine and personalized healthcare further drives the demand for sophisticated diagnostic tests that can provide detailed insights into pathogen characteristics and patient-specific responses, facilitating more effective treatment strategies.
Moreover, the increasing healthcare expenditure and the expansion of healthcare infrastructure in the United States contribute to the growth of the HAD testing market. The government and private sector investments in modernizing hospitals and healthcare facilities, coupled with the rising awareness about HAIs among healthcare providers and patients, have led to the implementation of robust infection control programs. These programs prioritize regular screening and monitoring of patients, healthcare workers, and hospital environments for potential infection sources. The COVID-19 pandemic has further underscored the importance of infection control and prevention, leading to heightened vigilance, and testing for various pathogens, including those responsible for HAIs. As hospitals and healthcare systems continue to prioritize patient safety and infection control, the demand for reliable and efficient HAD testing solutions is expected to remain strong.
United States Hospital Acquired Disease Testing Industry Segmentation:
The market report offers a comprehensive analysis of the segments, highlighting those with the largest United States hospital acquired disease testing market share. It includes forecasts for the period 2024-2032 and historical data from 2018-2023 for the following segments.
The report has segmented the market into the following categories:
Breakup by Indication:
- UTI (Urinary Tract Infection)
- SSI (Surgical Site Infection)
- Pneumonia
- Bloodstream Infections
- MRSA (Methicillin-Resistant Staphylococcus Aureus)
- Others
Breakup by Region:
- Northeast
- Midwest
- South
- West
Competitive Landscape:
The competitive landscape of the industry has also been examined along with the profiles of the key players.
Key highlights of the Report:
- Market Performance (2018-2023)
- Market Outlook (2024-2032)
- COVID-19 Impact on the Market
- Porter’s Five Forces Analysis
- Strategic Recommendations
- Historical, Current and Future Market Trends
- Market Drivers and Success Factors
- SWOT Analysis
- Structure of the Market
- Value Chain Analysis
- Comprehensive Mapping of the Competitive Landscape
Note: If you need specific information that is not currently within the scope of the report, we can provide it to you as a part of the customization.
Ask analyst for your customized sample: https://www.imarcgroup.com/request?type=report&id=2986&flag=F
Browse more research report:
About Us:
IMARC Group is a leading market research company that offers management strategy and market research worldwide. We partner with clients in all sectors and regions to identify their highest-value opportunities, address their most critical challenges, and transform their businesses.
IMARC’s information products include major market, scientific, economic and technological developments for business leaders in pharmaceutical, industrial, and high technology organizations. Market forecasts and industry analysis for biotechnology, advanced materials, pharmaceuticals, food and beverage, travel and tourism, nanotechnology and novel processing methods are at the top of the company’s expertise.
Contact Us:
IMARC Group
134 N 4th St. Brooklyn, NY 11249, USA
Email: [email protected]
Tel No:(D) +91 120 433 0800
United States: +1-631-791-1145